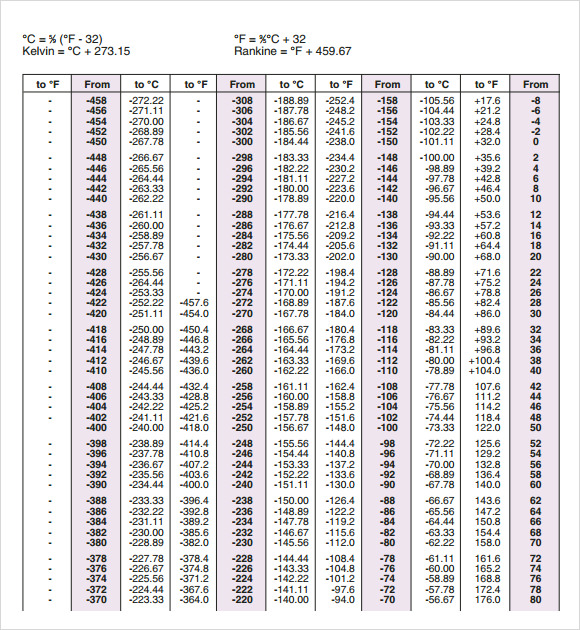
The specific heats of a few selected elements at 25 deg. 
The thermal capacity of materials changes slightly with temperature primarily due to changes in density, and very dramatically at phase transitions, such as ice melting and water boiling. The entire energy could be transferred to the water in one millisecond at a rate of 4184 watts to produce the same temperature rise of one deg. That is 4.184 Joules or 4.184 Watt-seconds. This is a measure of how many calories are needed to raise 1.0 gram of the material one degree Centigrade. 
Materials have a property called thermal capacity or specific heat. When measuring heating effects, a unit of thermal energy known as a calorie is used. So the power P in watts (W) is equal to the power P in BTUs per hour (BTU/hr) times 0.29307107 and is equal to the power P in BTUs per hour (BTU/hr) divided by 3. The energy is usually defined as Joules therefore, one Joule per second is one Watt. c Specific heat of the substance and m Mass. 1 cal 4.184 J 1 J 1 Ws (1 Ws) (1/3600 h/s) 2.78 10-4 Wh 2. Temperature is a physical quantity that expresses hot and cold. This is used frequently for observations at high-energy, be it for solar or X-ray astronomy. Formula: W C 1.16194 This formula for to heat 1 kilogram of water in 1 hour by 1 degree. The temperatureenergy () equivalency allows conversion between temperature and its equivalent in energy (i.e., the temperature multiplied by the Boltzmann constant), usually expressed in electronvolts. In order to convert Watt units into electricity units, one should use the power of. Power in Watts with which we heat the substance. Convert Celsius to Watts Temperature, C Power, Watt With this online calculator you can convert Celsius to Watt. The unit of 1 watt corresponds to the energy transfer rate of 1 Joule. How is the measurement Watts related to temperature in terms of energy?Ī Watt is a unit of power, defined as the rate of energy transferred. To calculate heat (actually, temperature change) from watts applied to a substance, use the formula: T (t × )/ (c × m), where: T Temperature change experienced by the substance t Time during which we apply the heat.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
